  
CATALYSIS OR COLLOID CHEMISTRY THEIR RELEVANT APPARATUS B01J- CHEMICAL OR PHYSICAL PROCESSES, e.g.B01- PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL.C01G- COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F. 
Application granted granted Critical Publication of US5207999A publication Critical patent/US5207999A/en Anticipated expiration legal-status Critical Status Expired - Fee Related legal-status Critical Current Links Assignors: BURK, ROBERT C., ZAWIDZKI, TADEUSZ W., GARRATT, DENNIS G. Google has not performed a legal analysis and makes no representation as to the accuracy of the date listed.) Filing date Publication date Priority to US07/744,413 priority Critical patent/US5207999A/en Application filed by Cameco Corp filed Critical Cameco Corp Priority to AU24390/92A priority patent/AU2439092A/en Priority to DE69216241T priority patent/DE69216241T2/en Priority to DK92917553.7T priority patent/DK0598801T3/en Priority to AT92917553T priority patent/ATE146761T1/en Priority to CA002115576A priority patent/CA2115576C/en Priority to PCT/CA1992/000352 priority patent/WO1993003997A1/en Priority to EP92917553A priority patent/EP0598801B1/en Assigned to CAMECO CORPORATION reassignment CAMECO CORPORATION ASSIGNMENT OF ASSIGNORS INTEREST. Original Assignee Cameco Corp Priority date (The priority date is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation or warranty as to the accuracy of the list.) Zawidzki Andrzej Huczko Current Assignee (The listed assignees may be inaccurate. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.) Expired - Fee Related Application number US07/744,413 Inventor Robert C. Google Patents Generation of fluorine via thermal plasma decomposition of metal fluorideĭownload PDF Info Publication number US5207999A US5207999A US07/744,413 US74441391A US5207999A US 5207999 A US5207999 A US 5207999A US 74441391 A US74441391 A US 74441391A US 5207999 A US5207999 A US 5207999A Authority US United States Prior art keywords plasma metal fluorine fluoride uranium Prior art date Legal status (The legal status is an assumption and is not a legal conclusion. Google Patents US5207999A - Generation of fluorine via thermal plasma decomposition of metal fluoride Some safety recommendations are included.US5207999A - Generation of fluorine via thermal plasma decomposition of metal fluoride General characteristics of the fluorocarbons, and some specific fluorocarbon materials are described. Materials of construction useful in handling fluorine are given, with indications of their limitations as to concentration, heat and thickness and specific recommendations on general design factors are itemized. The heat of reaction of fluorine is much greater than for oxygen or chlorine, and while metals that resist oxidation also may resist fluorine, contaminants, particularly organic materials, may react so rapidly as to initiate ignition of backing metal. 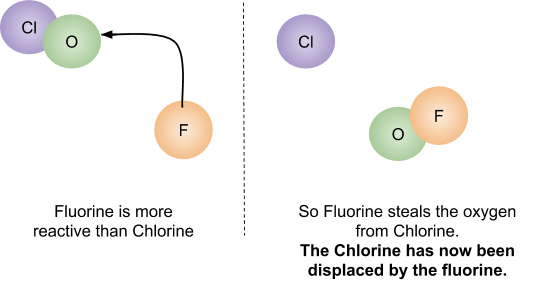
While fluorine is near oxygen and chlorine in the periodic table its 2.85 electronegativity is the highest known and makes it tend to displace them from their compounds. Because it tends to displace other elements and because no other element is sufficiently reactive to displace fluorine, fluorine compounds become chemically inert. The extreme reactivity of fluorine paradoxically brings to its compounds their most attractive properties-resistance to chemical attack. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
